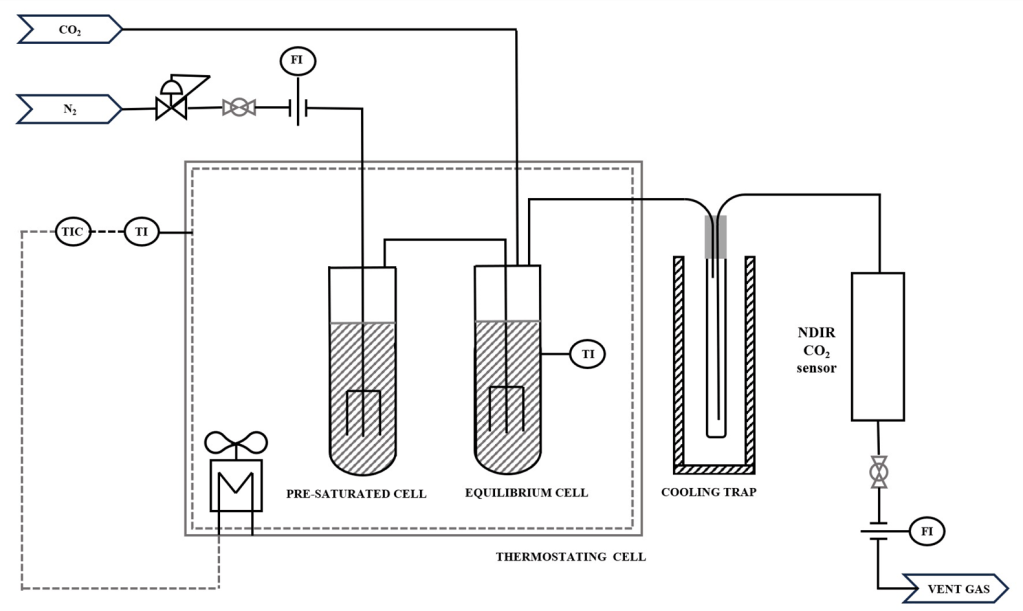
Saturator
Using a gas saturator in which an inert gas stream is introduced through a very thin capillary to obtain very small gas bubbles and equilibrium saturation of the gas stream, we are able to measure a variety of properties like
- gas solubilities
- activity coefficients at infinite dilution
- vapor pressure and VLE of high boiling mixtures (all condensables leaving the saturator are captured by a cooling trap and can be analysed

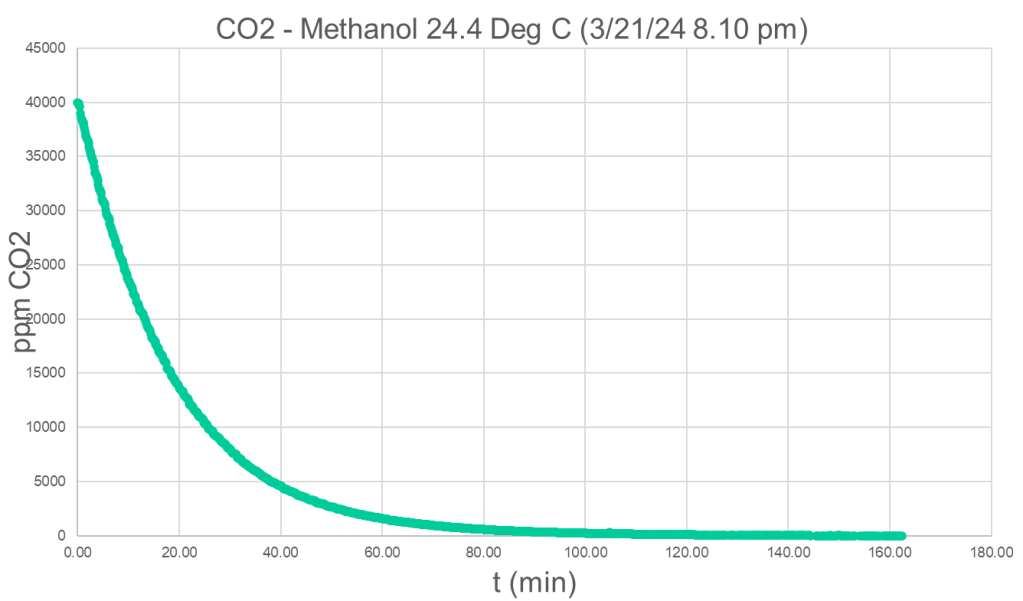
Different detectors are used to measure the concentration of the volatile component in the off-gas. In case of CO2, we modified commercial spectroscopic NDIR sensors. The following plot shows the decline of CO2 in the off-gas of a CO2 in methanol solution as function of time.
Using a logarithmic plot, the Henry constant can be calculated from the slope of the line:
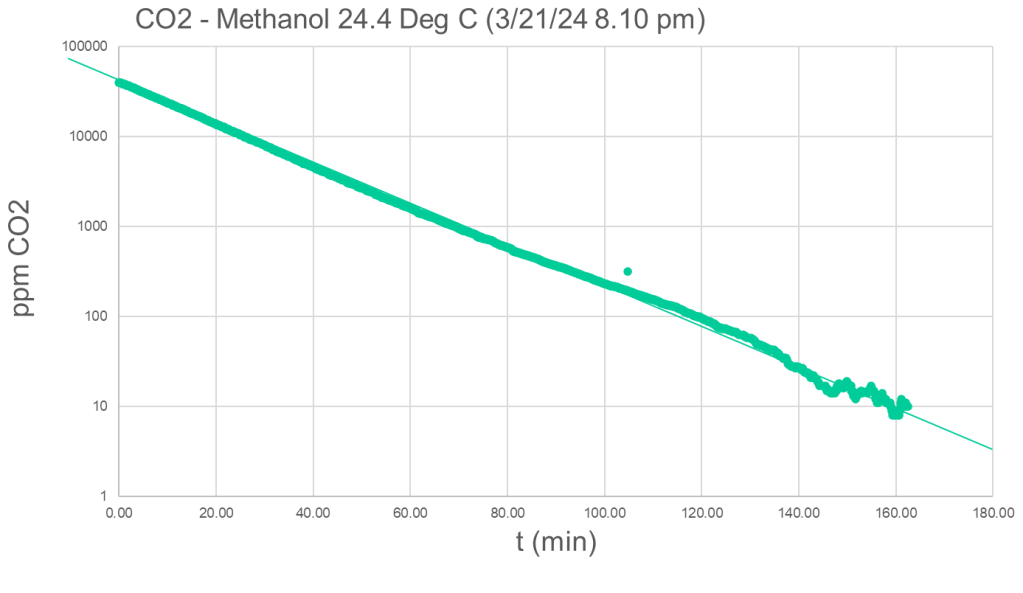
The slight bend in the curve results from room temperature change as this first proof-of-concept experiment was not conducted in a thermostat. We find nearly perfect linearity between CO2 partial pressures of 1 to 4000 Pa over more than 3 orders of magnitude.
Current work aims to expand this technique by evaluating NDIR sensors for different IR bands as well as a variety of electrochemical gas and VOC detectors.
The inert gas stripper is also used for measurement of pure component vapor pressures of heavy-boilers and VLE of heavy boiling mixtures.
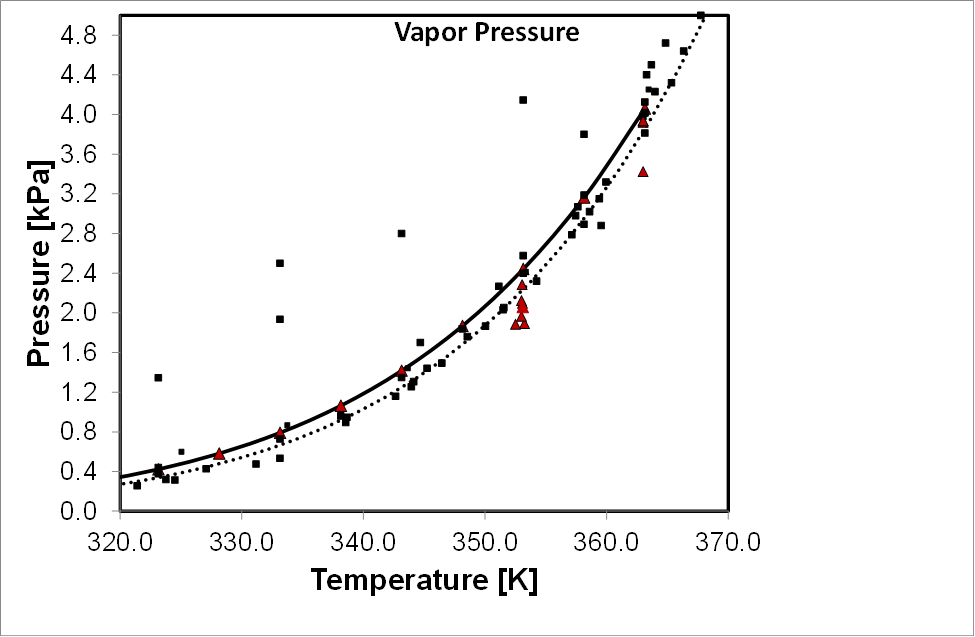
Vapor pressure data for monoethanolamine, red points are our measurements, calculated curves are included from DIPPR (via Aspen Plus, solid line) and the DDB (dotted line).